Engineering Simulation for Medical Device Design and Performance
Design, simulate, and validate medical devices with advanced engineering tools for structural reliability, thermal performance, electromagnetics, and regulatory compliance.

Engineering Capabilities for Medical Devices
Advanced simulation technologies to design, validate, and optimize medical devices for performance, safety, and regulatory compliance.

Structural Reliability of Medical Devices
Evaluate stress, fatigue, and durability of device assemblies to ensure performance and patient safety under real-world conditions.

Thermal Management for Medical Electronics
Simulate heat dissipation and cooling in handheld, wearable, and diagnostic devices to ensure reliability and user safety.

Electromagnetic and Wireless Performance
Analyze EMC/EMI behavior, antennas, and wireless communication to ensure compliance and reliable device operation.

Embedded Systems and Device Behavior
Model electromechanical systems, sensors, and control logic to simulate device behavior and performance before prototyping.
Manufacturability and Injection Molding
Validate plastic components and optimize manufacturing processes, including injection molding and production constraints.
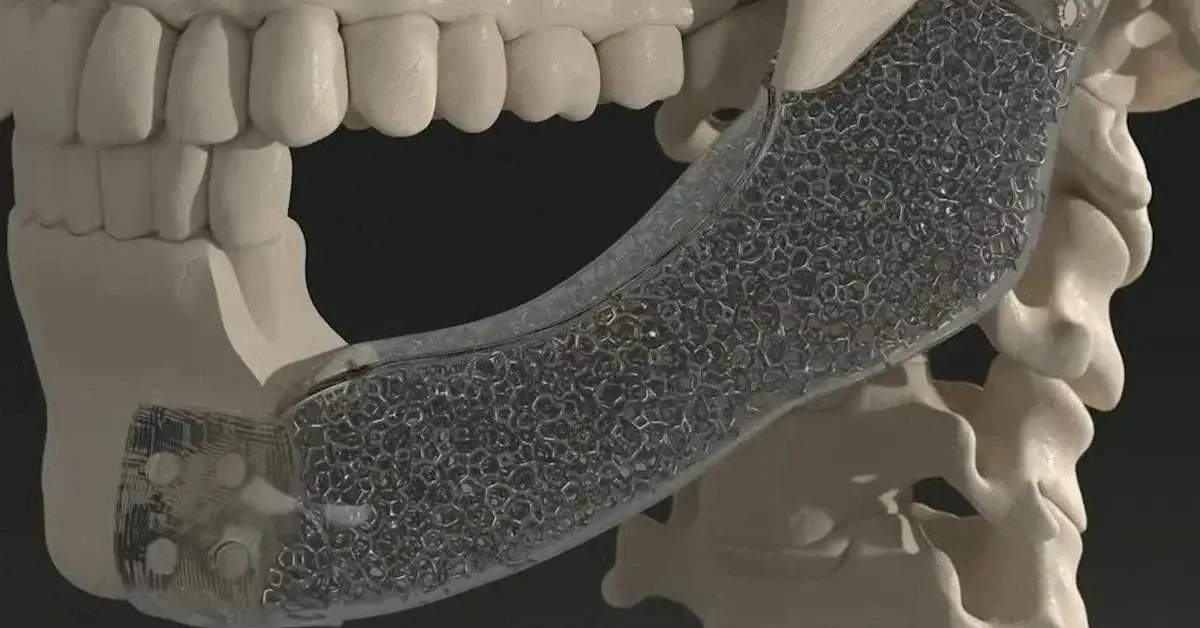
Additive Manufacturing for Medical Devices
Design and simulate 3D-printed components, optimizing lightweight structures and complex geometries for medical applications.

Digital Thread and Regulatory Traceability
Connect design, simulation, and manufacturing data to ensure traceability, compliance, and lifecycle management for regulated products.
Medical Device Applications
Simulation-driven approaches to design, validate, and optimize medical devices across performance, safety, and regulated development workflows.
Implantable Devices and Structural Performance
Simulate stress, fatigue, and long-term durability of implants to ensure reliability and patient safety under physiological conditions.
Wearable and Portable Medical Devices
Design compact, lightweight devices while validating ergonomics, thermal behavior, and real-world performance.
Medical Imaging and Diagnostic Equipment
Optimize performance and reliability of imaging and diagnostic systems, including thermal behavior, structural integrity, and system interactions.
Connected Medical Devices and Wireless Systems
Design and validate wireless-enabled devices, ensuring reliable communication, EMC compliance, and secure data transmission.
Electromechanical Medical Systems
Simulate interactions between mechanical components, sensors, and control systems in complex medical devices.
Regulated Product Development and Traceability
Ensure compliance with regulatory standards by connecting design, simulation, and manufacturing data across the full product lifecycle.
Key Technologies for Medical Device Engineering
Explore the engineering technologies used to design, simulate, and validate medical devices across structural performance, thermal behavior, electronics, and regulated development workflows.

Structural Analysis and Multiphysics Validation
Evaluate structural integrity, fatigue, and multiphysics behavior in medical devices using advanced simulation tools.
Know Structural Simulation →
Thermal and Fluid Simulation
Simulate heat transfer and fluid behavior in medical devices, including cooling of electronics and flow in diagnostic systems.
Explore Thermal Simulation →
Electromagnetic and EMC Simulation
Analyze EMC/EMI performance, antennas, and wireless communication in medical electronics to ensure compliance and reliability.
View EMC & RF Tools →
Embedded Systems and Control Modeling
Model sensors, control systems, and electromechanical behavior in medical devices for performance validation.
Explore Control Systems →
Manufacturing and Injection Molding
Validate manufacturability and optimize plastic components using injection molding and production simulation tools.
View Manufacturing Tools →
Additive Manufacturing for Medical Devices
Design and simulate 3D-printed medical components, enabling complex geometries and optimized structures.
Explore Additive Manufacturing →Medical Device Development Workflow
Modern medical devices are developed through integrated workflows combining design, simulation, electronics, and regulatory validation.
Concept Design
Create device components and assemblies using advanced CAD tools, enabling rapid prototyping and early-stage design validation.
Know Solid Edge →
Structural Simulation and Optimization
Evaluate structural performance, fatigue, and lightweight design using simulation-driven engineering approaches for medical devices.
Explore Structural Simulation →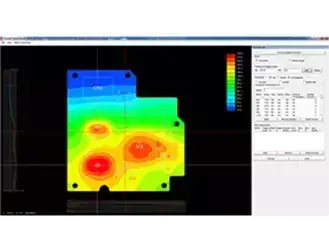
Multiphysics and Thermal Simulation
Simulate thermal behavior, fluid flow, and coupled physics interactions affecting device performance and safety.
View Multiphysics Simulation →
Electronics and Embedded Systems
Model sensors, control systems, and embedded electronics to validate device behavior and ensure reliable operation.
Explore Embedded Systems →
Manufacturing and Regulatory Validation
Validate manufacturability and ensure compliance through simulation of production processes and lifecycle traceability.
View Manufacturing Tools →Medical Device Engineering Software FAQ
Medical device engineering uses simulation tools for structural analysis, thermal management, electromagnetics, and system modeling. These tools help validate performance, safety, and compliance before physical prototyping.
Structural simulation tools evaluate stress, fatigue, and long-term durability of device components. This is especially important for implants and load-bearing devices to ensure patient safety and regulatory compliance.
Thermal simulation is used to analyze heat generation and dissipation in devices such as wearables, diagnostic equipment, and electronics. It ensures safe operating temperatures and reliable performance.
Electromagnetic simulation tools analyze EMC/EMI behavior, antenna performance, and wireless communication. This ensures devices operate reliably without interference and meet regulatory standards.
Simulation provides validated data for design verification, risk analysis, and documentation. It helps meet regulatory requirements by improving traceability and reducing reliance on physical testing.
Simulation enables engineers to test performance, optimize designs, and identify issues early in development. This reduces costs, accelerates time-to-market, and improves overall device reliability.
Ready to Validate Your Medical Devices with Confidence?
Leverage simulation-driven engineering to improve device performance, ensure regulatory compliance, and reduce development risk across the entire product lifecycle.



